State health officials call COVID-19 vaccine ‘safe and effective’
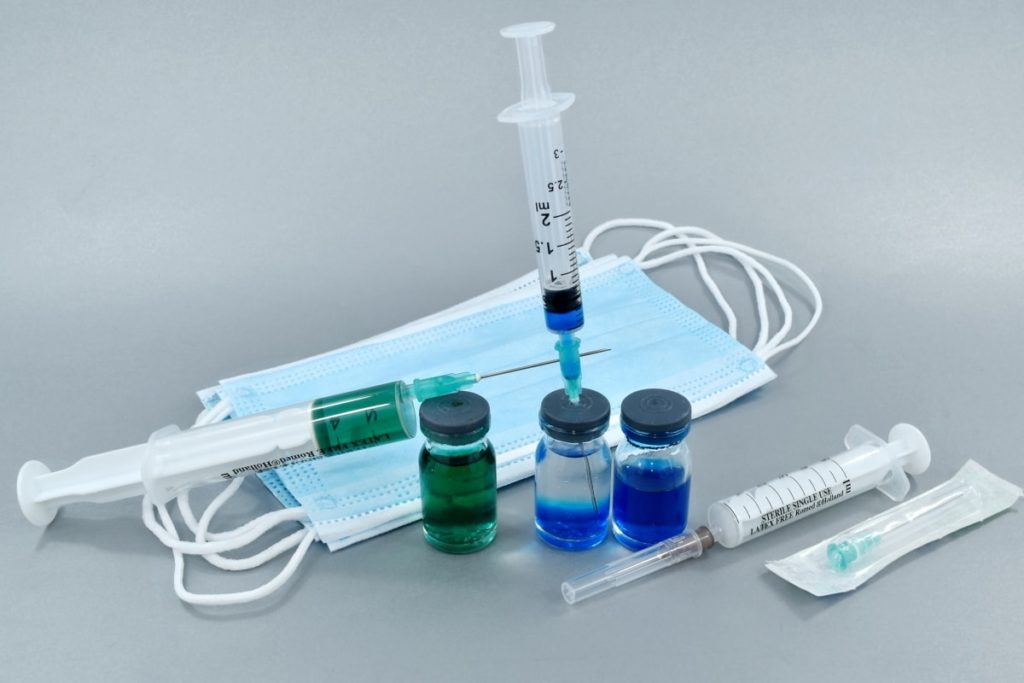
Once the U.S. Food and Drug Administration gives its approval, which is expected Thursday, hospitals could begin vaccinating staff within 24 to 72 hours, Health Department officials said
New Jersey health officials said they are confident that the COVID-19 vaccine is safe and effective, based on the information they have reviewed, and hope hospitals could start immunizing the most at-risk individuals within a few days of it receiving federal approval.
Gov. Phil Murphy had said the vaccine program would not begin without a state-level review of clinical trial data in addition to the national Food and Drug Administration approval. The FDA is scheduled to meet Thursday to determine if Pfizer’s vaccine should receive an emergency use authorization, which would enable coronavirus immunizations to begin nationwide.
On Monday, Murphy and Dr. Edward Lifshitz, director of the state Health Department’s communicable disease system, suggested this state review — which the governor has frequently likened to “kicking the tires” on the vaccine — is essentially complete. That means that once FDA approval is secured, hospitals could begin vaccinating staff within 24 to 72 hours, department officials said.








Come on now – How quickly everyone forgets about the measles outbreak we had 2 years ago? We’ve have a vaccine for that for like 50 years and we almost eradicated until a bunch of people saw some crazy videos on YouTube.
Any vaccine will only work as well as people are willing to take it!